Introduction to Population Pharmacokinetic / Pharmacodynamic Analysis with Nonlinear Mixed Effects Models
Nonfiction, Health & Well Being, Medical, Reference, Biostatistics, Medical Science, Pharmacology, Science & Nature, Mathematics, Statistics| Author: | Joel S. Owen, Jill Fiedler-Kelly | ISBN: | 9781118784617 |
| Publisher: | Wiley | Publication: | June 19, 2014 |
| Imprint: | Wiley | Language: | English |
| Author: | Joel S. Owen, Jill Fiedler-Kelly |
| ISBN: | 9781118784617 |
| Publisher: | Wiley |
| Publication: | June 19, 2014 |
| Imprint: | Wiley |
| Language: | English |
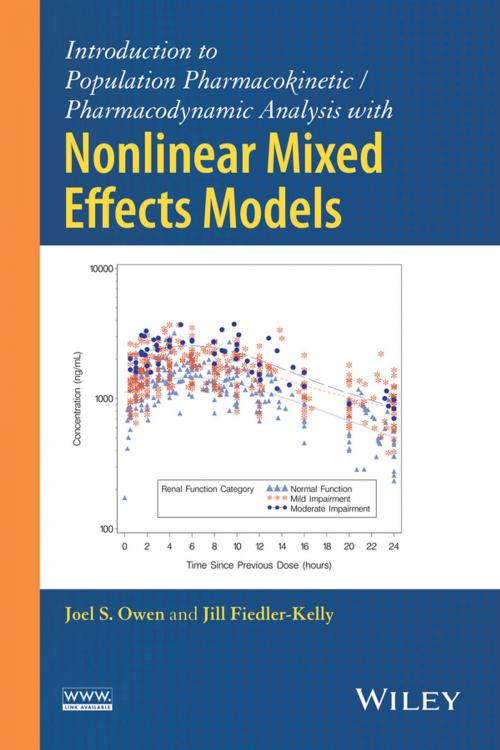
This book provides a user-friendly, hands-on introduction to the Nonlinear Mixed Effects Modeling (NONMEM) system, the most powerful tool for pharmacokinetic / pharmacodynamic analysis.
• Introduces requisite background to using Nonlinear Mixed Effects Modeling (NONMEM), covering data requirements, model building and evaluation, and quality control aspects
• Provides examples of nonlinear modeling concepts and estimation basics with discussion on the model building process and applications of empirical Bayesian estimates in the drug development environment
• Includes detailed chapters on data set structure, developing control streams for modeling and simulation, model applications, interpretation of NONMEM output and results, and quality control
• Has datasets, programming code, and practice exercises with solutions, available on a supplementary website
This book provides a user-friendly, hands-on introduction to the Nonlinear Mixed Effects Modeling (NONMEM) system, the most powerful tool for pharmacokinetic / pharmacodynamic analysis.
• Introduces requisite background to using Nonlinear Mixed Effects Modeling (NONMEM), covering data requirements, model building and evaluation, and quality control aspects
• Provides examples of nonlinear modeling concepts and estimation basics with discussion on the model building process and applications of empirical Bayesian estimates in the drug development environment
• Includes detailed chapters on data set structure, developing control streams for modeling and simulation, model applications, interpretation of NONMEM output and results, and quality control
• Has datasets, programming code, and practice exercises with solutions, available on a supplementary website